목차
1. 서 론
2. 실험방법
2.1 LiMn2-δTiδO4(0≤δ≤0.2)의 합성
2.2 LiMn2-δTiδO4(0≤δ≤0.2)의 결정구조 정밀화
3. 결과 및 고찰
4. 결 론
5. 참 고 문 헌
2. 실험방법
2.1 LiMn2-δTiδO4(0≤δ≤0.2)의 합성
2.2 LiMn2-δTiδO4(0≤δ≤0.2)의 결정구조 정밀화
3. 결과 및 고찰
4. 결 론
5. 참 고 문 헌
본문내용
e 3 Crystallographic parameter of Li0.05Mn2O4("λ-MnO2") with a = 8.0445Å and Space Group = Fd3m
Table 4 Refined atomic parameters of LiMn2-δTiδO4(0≤δ≤0.2) sintered at 850℃
Table 5 Refined atomic parameters of LiMn2-δTiδO4(0≤δ≤0.2) sintered at 1100℃
FIGURE CAPTION
Fig. 1 XRD patterns of Li(Mn1-δTiδ)2O4 system sintered at 850℃
(a) δ=0 (b) δ=0.05 (c) δ=0.1 (d) δ=0.2
Fig. 2 XRD patterns of Li(Mn1-δTiδ)2O4 system sintered at 1100℃
(a) δ=0 (b) δ=0.05 (c) δ=0.1 (d) δ=0.2
Fig. 3 X-ray Rietveld refinement of LiMn2O4 sintered at 850℃
Fig. 4 X-ray Rietveld refinement of LiMn2O4 sintered at 1100℃
Fig. 5 X-ray Rietveld refinement of Li(Mn0.80Ti0.20)2O4 sintered at 850℃
Fig. 6 X-ray Rietveld refinement of Li(Mn0.80Ti0.20)2O4 sintered at 1100℃
Fig. 7 The variation of the lattice parameters with titanium content
Fig. 8 The variation of x-parameter on 32e-site with titanium content
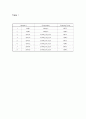
Table 1
Sample #
Composition
Sintering Temp.
1
LM01
LiMn2O4
850℃
2
LM02
LiMn2O4
1100℃
3
LMT11
Li(Mn0.95Ti0.05)2O4
850℃
4
LMT12
Li(Mn0.95Ti0.05)2O4
1100℃
5
LMT21
Li(Mn0.90Ti0.10)2O4
850℃
6
LMT22
Li(Mn0.90Ti0.10)2O4
1100℃
7
LMT31
Li(Mn0.80Ti0.20)2O4
850℃
8
LMT32
Li(Mn0.80Ti0.20)2O4
1100℃
Table 2
Li : 8a 0, 0, 0 ; 3/4, 1/4, 3/4
M(Mn1-δTiδ) : 16d 5/8, 5/8, 5/8 ; 3/8, 7/8, 1/8 ; 7/8, 1/8, 3/8 ; 1/8, 3/8, 7/8
O : 32e x, x, x ; -x, -x+1/2, x+1/2 ; -x+1/2, x+1/2, -x ; x+1/2, -x, -x+1/2
x+3/4, x+1/4, -x+3/4 ; -x+1/4, -x+1/4, -x+1/4
x+1/4, -x+3/4, x+3/4 ; -x+3/4, x+3/4, x+3/4
Table 3
Atom
Position
x
y
z
β11
β12
Occupancy
Li
8a
0
0
0
0.39
-
0.05
Mn
16d
0.625
0.625
0.625
0.39
-
1.0
O
32e
0.3881
0.3881
0.3881
0.45
0.03
1.0
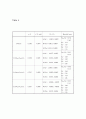
Table 4
a/Å
x (O-site)
β11, β12
Residual Index
LiMn2O4
8.2363
0.3876
Li(8a) : -0.0013, 0.0097
M(16d): 0.0027, 0.0004
O(32e): 0.0114, 0.0003
Rwp, Rp= 15.70,
11.60
Re = 5.72
RI = 2.92
RF = 2.16
S = 2.74
Li(Mn0.95Ti0.05)2O4
8.2459
0.3881
Li(8a) : 0.0157, 0.0025
M(16d): 0.0014, 0.0001
O(32e): 0.0152, 0.0002
Rwp, Rp= 19.81,
13.16
Re = 5.66
RI = 5.03
RF = 4.43
S = 3.50
Li(Mn0.90Ti0.10)2O4
8.2522
0.3885
Li(8a) : 0.0087, 0.0050
M(16d): 0.0009, 0.0002
O(32e): 0.0167, 0.0004
Rwp, Rp= 19.60,
14.18
Re = 5.66
RI = 5.31
RF = 4.44
S = 3.46
Li(Mn0.80Ti0.20)2O4
8.2857
0.3890
Li(8a) : -0.0056, -0.0078
M(16d): 0.0009, -0.0003
O(32e): 0.0131, 0.0016
Rwp, Rp= 15.85
11.41
Re = 5.60
RI = 4.82
RF = 3.85
S = 2.83
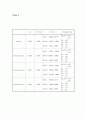
Table 5
a/Å
x (O-site)
β11, β12
Residual Index
LiMn2O4
8.2383
0.3858
Li(8a) : -0.0036, 0.0139
M(16d): 0.0026, -0.0002
O(32e): 0.0167, 0.0001
Rwp, Rp= 16.29
11.31
Re = 6.03
RI = 2.98
RF = 3.15
S = 2.70
Li(Mn0.95Ti0.05)2O4
8.2506
0.3875
Li(8a) : 0.0164, -0.0059
M(16d):-0.0010, -0.0002
O(32e): 0.0463, 0.0023
Rwp, Rp= 16.43
11.15
Re = 5.85
RI = 3.69
RF = 3.12
S = 2.81
Li(Mn0.90Ti0.10)2O4
8.2632
0.3845
Li(8a) : 0.0156, -0.0051
M(16d): 0.0004, 0.0000
O(32e): 0.01481, 0.0003
Rwp, Rp= 16.01
11.09
Re = 5.73
RI = 3.19
RF = 2.73
S = 2.79
Li(Mn0.80Ti0.20)2O4
8.2880
0.3884
Li(8a) : -0.0026, 0.0116
M(16d): 0.0000, 0.0005
O(32e): 0.0017, 0.0025
Rwp, Rp= 14.28
10.30
Re = 5.68
RI = 2.20
RF = 1.74
S = 2.51
Table 4 Refined atomic parameters of LiMn2-δTiδO4(0≤δ≤0.2) sintered at 850℃
Table 5 Refined atomic parameters of LiMn2-δTiδO4(0≤δ≤0.2) sintered at 1100℃
FIGURE CAPTION
Fig. 1 XRD patterns of Li(Mn1-δTiδ)2O4 system sintered at 850℃
(a) δ=0 (b) δ=0.05 (c) δ=0.1 (d) δ=0.2
Fig. 2 XRD patterns of Li(Mn1-δTiδ)2O4 system sintered at 1100℃
(a) δ=0 (b) δ=0.05 (c) δ=0.1 (d) δ=0.2
Fig. 3 X-ray Rietveld refinement of LiMn2O4 sintered at 850℃
Fig. 4 X-ray Rietveld refinement of LiMn2O4 sintered at 1100℃
Fig. 5 X-ray Rietveld refinement of Li(Mn0.80Ti0.20)2O4 sintered at 850℃
Fig. 6 X-ray Rietveld refinement of Li(Mn0.80Ti0.20)2O4 sintered at 1100℃
Fig. 7 The variation of the lattice parameters with titanium content
Fig. 8 The variation of x-parameter on 32e-site with titanium content
Table 1
Sample #
Composition
Sintering Temp.
1
LM01
LiMn2O4
850℃
2
LM02
LiMn2O4
1100℃
3
LMT11
Li(Mn0.95Ti0.05)2O4
850℃
4
LMT12
Li(Mn0.95Ti0.05)2O4
1100℃
5
LMT21
Li(Mn0.90Ti0.10)2O4
850℃
6
LMT22
Li(Mn0.90Ti0.10)2O4
1100℃
7
LMT31
Li(Mn0.80Ti0.20)2O4
850℃
8
LMT32
Li(Mn0.80Ti0.20)2O4
1100℃
Table 2
Li : 8a 0, 0, 0 ; 3/4, 1/4, 3/4
M(Mn1-δTiδ) : 16d 5/8, 5/8, 5/8 ; 3/8, 7/8, 1/8 ; 7/8, 1/8, 3/8 ; 1/8, 3/8, 7/8
O : 32e x, x, x ; -x, -x+1/2, x+1/2 ; -x+1/2, x+1/2, -x ; x+1/2, -x, -x+1/2
x+3/4, x+1/4, -x+3/4 ; -x+1/4, -x+1/4, -x+1/4
x+1/4, -x+3/4, x+3/4 ; -x+3/4, x+3/4, x+3/4
Table 3
Atom
Position
x
y
z
β11
β12
Occupancy
Li
8a
0
0
0
0.39
-
0.05
Mn
16d
0.625
0.625
0.625
0.39
-
1.0
O
32e
0.3881
0.3881
0.3881
0.45
0.03
1.0
Table 4
a/Å
x (O-site)
β11, β12
Residual Index
LiMn2O4
8.2363
0.3876
Li(8a) : -0.0013, 0.0097
M(16d): 0.0027, 0.0004
O(32e): 0.0114, 0.0003
Rwp, Rp= 15.70,
11.60
Re = 5.72
RI = 2.92
RF = 2.16
S = 2.74
Li(Mn0.95Ti0.05)2O4
8.2459
0.3881
Li(8a) : 0.0157, 0.0025
M(16d): 0.0014, 0.0001
O(32e): 0.0152, 0.0002
Rwp, Rp= 19.81,
13.16
Re = 5.66
RI = 5.03
RF = 4.43
S = 3.50
Li(Mn0.90Ti0.10)2O4
8.2522
0.3885
Li(8a) : 0.0087, 0.0050
M(16d): 0.0009, 0.0002
O(32e): 0.0167, 0.0004
Rwp, Rp= 19.60,
14.18
Re = 5.66
RI = 5.31
RF = 4.44
S = 3.46
Li(Mn0.80Ti0.20)2O4
8.2857
0.3890
Li(8a) : -0.0056, -0.0078
M(16d): 0.0009, -0.0003
O(32e): 0.0131, 0.0016
Rwp, Rp= 15.85
11.41
Re = 5.60
RI = 4.82
RF = 3.85
S = 2.83
Table 5
a/Å
x (O-site)
β11, β12
Residual Index
LiMn2O4
8.2383
0.3858
Li(8a) : -0.0036, 0.0139
M(16d): 0.0026, -0.0002
O(32e): 0.0167, 0.0001
Rwp, Rp= 16.29
11.31
Re = 6.03
RI = 2.98
RF = 3.15
S = 2.70
Li(Mn0.95Ti0.05)2O4
8.2506
0.3875
Li(8a) : 0.0164, -0.0059
M(16d):-0.0010, -0.0002
O(32e): 0.0463, 0.0023
Rwp, Rp= 16.43
11.15
Re = 5.85
RI = 3.69
RF = 3.12
S = 2.81
Li(Mn0.90Ti0.10)2O4
8.2632
0.3845
Li(8a) : 0.0156, -0.0051
M(16d): 0.0004, 0.0000
O(32e): 0.01481, 0.0003
Rwp, Rp= 16.01
11.09
Re = 5.73
RI = 3.19
RF = 2.73
S = 2.79
Li(Mn0.80Ti0.20)2O4
8.2880
0.3884
Li(8a) : -0.0026, 0.0116
M(16d): 0.0000, 0.0005
O(32e): 0.0017, 0.0025
Rwp, Rp= 14.28
10.30
Re = 5.68
RI = 2.20
RF = 1.74
S = 2.51
키워드
추천자료
 다수인공동의 의사결정과정에서의 의결행위와 형법해석학적 의미
다수인공동의 의사결정과정에서의 의결행위와 형법해석학적 의미 국내외 토목구조물 중 Best & Worst 구조물을 결정하고, 구조와 특징 등을 설명하라.
국내외 토목구조물 중 Best & Worst 구조물을 결정하고, 구조와 특징 등을 설명하라. 고려가요(고려속요) 처용가의 유래와 배경설화, 고려가요(고려속요) 처용가의 원문, 고려가요...
고려가요(고려속요) 처용가의 유래와 배경설화, 고려가요(고려속요) 처용가의 원문, 고려가요... 처용가(고려가요, 고려속요)의 성격, 처용가(고려가요, 고려속요)의 형성과정과 처용가(고려...
처용가(고려가요, 고려속요)의 성격, 처용가(고려가요, 고려속요)의 형성과정과 처용가(고려... [화학][화학적][모발][물][내층세포][단백질합성][유전자][생명]모발의 화학적 성질, 물의 화...
[화학][화학적][모발][물][내층세포][단백질합성][유전자][생명]모발의 화학적 성질, 물의 화... 정책결정의 개념구조와 정책결정과정
정책결정의 개념구조와 정책결정과정 가계도의 정의와 활용시의 장점을 기술하고, 자신의 가계도를 작성하여 그 가계도를 해석 - ...
가계도의 정의와 활용시의 장점을 기술하고, 자신의 가계도를 작성하여 그 가계도를 해석 - ... [산업조직의 진화] 수평적-수직적 조직의 진화(수직적 산업구조와 수직적 산업구조의 장점, ...
[산업조직의 진화] 수평적-수직적 조직의 진화(수직적 산업구조와 수직적 산업구조의 장점, ... [무기화학 및 분석화학 실험] 04[결과]. MCM-41 합성 및 분석 : Mesoporous Material중 하나...
[무기화학 및 분석화학 실험] 04[결과]. MCM-41 합성 및 분석 : Mesoporous Material중 하나... [무기화학 및 분석화학 실험] 06[예비]. 나노사이즈의 SiO2 합성 및 분석 : 실리카(Silica)를...
[무기화학 및 분석화학 실험] 06[예비]. 나노사이즈의 SiO2 합성 및 분석 : 실리카(Silica)를... [어휘구조론] 조어법(파생법과 합성법)을 통한 예시
[어휘구조론] 조어법(파생법과 합성법)을 통한 예시 [무기화학실험] Macrocyclic Ligands and Their Metal Complexes : 킬레이트 반응을 통해 거...
[무기화학실험] Macrocyclic Ligands and Their Metal Complexes : 킬레이트 반응을 통해 거... [무기화학실험] 헥사아민코발트1 - 코발트(Ⅲ) 착화합물의 합성을 통한 착이온 조성의 제어 : ...
[무기화학실험] 헥사아민코발트1 - 코발트(Ⅲ) 착화합물의 합성을 통한 착이온 조성의 제어 : ... [정신분석이론]프로이드의 정신분석이론 - 성격의 구조(원초아,자아,초자아), 프로이트이론의...
[정신분석이론]프로이드의 정신분석이론 - 성격의 구조(원초아,자아,초자아), 프로이트이론의...